Case Report - (2024) Volume 9, Issue 12
Recurrent Transient Ischemic Attack in a Pediatric Female - A Case Report
2Second year medical student, California Northstate University College of Medicine, USA
3Assistant Professor, California Northstate University College of Medicine, USA, and Emergency Physic, USA
4Professor of Emergency Medicine and The Director of M4 Class, California Northstate University Colle, USA
Received Date: Nov 13, 2024 / Accepted Date: Nov 25, 2024 / Published Date: Dec 18, 2024
Copyright: ©2024 Leonard Ranasinghe, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Tu, S., Tanwar, A., Kouch, E., Ranasinghe, N., Ranasinghe, L. (2024). Recurrent Transient Ischemic Attack in a Pediatric Female - A Case Report. J Clin Rev Case Rep, 9(12), 01-04.
Abstract
Background: In adults, transient ischemic attack (TIA) is a risk factor for developing ischemic stroke within two days. While there is growing literature on TIA in adults and its epidemiology, there have been few cases that discuss the epidemiology and medical conditions associated with TIA in the pediatric age group.
Purpose: To discuss a novel case of pediatric TIA
Presentation: A 17-year-old morbidly obese female, who presented to the ED with intermittent left lower leg weakness for about 3 weeks then loss of left lower extremity strength for several minutes that now resolved. The patient had labs done and was imaged with a computed tomography (CT) scan.
Conclusion: This case report will provide insight to different medical conditions and risk factors that potentially led to a child developing a recurrent TIA.
Keywords
Transient Ischemic Attack, Pediatric, Case Report
Introduction
Transient ischemic attack (TIA) is a transient episode of neurologic dysfunction due to focal brain, spinal cord, or retinal ischemia, without acute infarction or tissue injury, typically lasting a few minutes to a few hours but may last up to 24 hours.
In a review article, Panuganti et al described the pathophysiology in that transient interruption of arterial blood flow to an area of the brain by a particular artery could be due to large artery atherothrombosis or small vessel ischemic diseases from hypertension or cardiac embolism [7]. Risk factors for TIA include family history of stroke or TIA, age above 55 years or older, male gender, high blood pressure, diabetes, imbalance in lipid profile, and tobacco smoking [3]. TIA is diagnosed after taking a thorough history and physical examination, and radiological tests such as computed tomography (CT) and magnetic resonance imaging (MRI) are important for evaluation [3]. In adults, one-third of the people with TIA can later have a recurrent TIA and one-third can have a stroke due to permanent nerve cell loss.
Studies have shown that those who have had a history of TIA have a significantly greater risk of impending ischemic stroke, the highest risk being in the first 48 hours. Therefore, heightened awareness, identification of persons at high risk, and timely management are extremely important. In adults with TIA, 1% to 10% will have a stroke within the next 48 hours, and up to 15% will have a stroke within 3 months; however, a recent study showed that in children, 16% developed radiological evidence of ischemic cerebral injury after a mean 4.5 year follow up period and 13% had a stroke after TIA. Risk factors for stroke after TIA in adults include presentation with motor symptoms, aphasia without motor symptoms, and signal changes consistent with ischemia on diffusion MRI [2,3,6,7,10]. In addition, Lehman’s study showed arteriopathy, female sex, and autoimmune disorders such as sickle cell disease significantly associated with stroke after TIA but does not show whether there is a significant association between symptom type and subsequent stroke [4].
While there is a wide variety of literature showing significant risk factors in predicting TIA in adults, very few studies have described it in a pediatric population (children 11-18). Thus, understanding the epidemiology and medical conditions associated with TIA in the pediatric and adolescent age group will allow for appropriate evaluation and better patient treatment. This report will investigate the uniqueness of presentation of TIA in this patient. We will be evaluating the patient’s symptoms, diagnosis, and progression then compare it to findings in literature regarding similar presentations. Since the annual incidence of TIA in children is 0.54 per 100,000, the ultimate goal is to highlight the uniqueness of this patient’s case and its relevance to evaluate future patients with similar presentations.
Materials and Methods
A review of a clinical case was conducted to examine transient ischemic attack. The clinical case was compiled by Dr. Nalin Ranasinghe.
Case Presentation
We present a case of a 17-year-old Caucasian American female, with a past medical history of essential hypertension, cerebral artery occlusion and infarction, hyperlipidemia, seasonal allergies, and morbid obesity, who came to the emergency department with a chief complaint of loss of left lower extremity strengthening for several minutes that has now been resolved. Two years ago, she had a cerebral artery occlusion with cerebral infarction and was then prescribed Coumadin. Over the last three weeks, the patient had been experiencing intermittent left lower leg weakness. Then, at 9:50am, she lost full strength of her left lower extremity. She denied previous surgery or trauma and any family history of stroke or TIA. Her daily medications were Apixaban 5 mg, Aspirin 81 mg, Atorvastatin 40 mg, Lisinopril 10 mg, Rivaroxaban 20 mg, and Warfarin 5 mg. She denied tobacco, alcohol, and drug use and denied sexual activity. Her review of systems was positive for neurological weakness, but the patient denied dizziness, tremors, seizures, facial asymmetry, speech difficulty, numbness, and headache.
On physical exam, her vitals were Temp 98.4°F, WT 316 lbs, BP 117/75, HR 81, RR 27 and SpO2 100%. The patient appeared well-developed with normal appearance and was not ill or toxic appearing, or diaphoretic. She was not in acute distress. On her head, eyes, ears, nose, and throat (HEENT) exam, extraocular movements were intact, conjunctivae was normal, and pupils were equal, round, and reactive to light. On her musculoskeletal (MSK) exam, there was normal range of motion and no rigidity, tenderness, or pedal edema. On neurological exam, there was no focal deficit present, and the patient was alert and oriented to person, place, and time. There was no cranial nerve deficit, sensory deficit, weakness or abnormal muscle tone. There was normal coordination and gait.
Lab results for CBC and CMP showed low MCV (80.0 fL) and MCH (25.6 pg), but the rest were unremarkable. Troponin I level (<3.0) was unremarkable. Her INR was 2.2, within therapeutic range for those taking Coumadin. ECG showed normal sinus rhythm with incomplete left bundle branch block. CT brain without contrast on 04/10/2022 demonstrated encephalomalacia with old infarct in the right MCA territory but no new large acute territorial infarction, no hemorrhage, and no midline shift or mass effect. She was later transferred from a local hospital to a Level 1 Pediatric Trauma Center for a brain MRI.
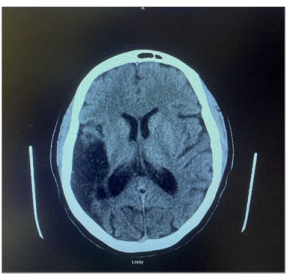
Figure 1: CT Brain Without Contrast on 04/10/2022 Demonstrated Encephalomalacia with Old Infarct in the Right Middle Cerebral Artery Territory but No New Large Acute Territorial Infarction, No Hemorrhage, and No Midline Shift or Mass Effect.
Discussion
This case illustrates that pediatric and adult patients may have similar risk factors for recurrent TIA. Despite being on coumadin and aspirin, the patient had a recurrent TIA. The patient’s comorbidities such as hypertension, hyperlipidemia, and morbid obesity as well as a significant past medical history of prior stroke and TIA most likely led to a recurrence of TIA. Additionally, it may also be possible this patient was not compliant with taking medications as prescribed contributing to her recurrent TIA episodes. High blood pressure and imbalance in lipid profile should be considered a greater threat for recurrent TIA in pediatric patients. Pediatric patients with multiple risk factors and history of stroke/TIA should be monitored, despite being on medication.
Demographics of TIA in Children
The annual incidence of hemorrhagic or ischemic stroke in children is 4.3 per 100,000 whereas for TIA, it is 0.54 per 100,000.
In a study that analyzed prevalence of and risk factors for children with TIA requiring hospitalization in the US from 2003-2009, researchers found that TIA was the primary diagnosis for 531 children and the median age was 13 years, and 48% were male [1]. In addition, TIAs were more common in adolescents; 67% occurred in those ages 11-18 years. Of those children, 20% had sickle cell disease, 11% had congenital heart disease, 12% had migraine, 1-% had moyamoya disease, and 4% had stroke [1]. Researchers noted that anemia, coagulopathy, diabetes, hypertension, and obesity were rare comorbid conditions, each noted in less than 6% of children. 217 (40.0%) had no risk factor identified [1]. A third of children who had confirmed arterial ischemic stroke (AIS) had a history of recent TIAs, and prior TIA is a strong risk factor for recurrent stroke in children [1]. Lehman’s study also showed that stroke occurred after TIA at a rate similar to that seen in adults, but the risk factors for stroke after TIA in children are different. Arteriopathy, female sex, and autoimmune disorders were significant risk factors [4].
Risk Factors of TIA
Nonmodifiable risk factors include age, sex, race, and significant family history. Well-documented and modifiable risk factors include tobacco, smoking, obesity, physical inactivity, cardiovascular, and lipid profile deranges such as coronary artery disease, myocardial infarction, valvular disease, atrial fibrillation, and diabetes mellitus. Potentially modifiable risk factors include history of migraine, obstructive sleep apnea, sleep patterns, and high-risk alcohol consumption [4].
Among the major, modifiable risk factors, high blood pressure is the most significant one for TIA and ischemic stroke [4,8,10]. Studies have shown a decrease in recurrence rates on antihypertensive interventions but some issues that arose include the variability of systolic BP and diastolic BP, technical errors, and criteria for initiating medication [4,7].
Secondary Recurrence of TIA
A TIA occurs when there is a transient block to the brain’s blood supply, the patient’s hypertension and high cholesterol levels may have caused the attack. However, this case is unusual due to the patient’s young age, medication history, and neurologic presentation that occurred 3 weeks prior to the attack. Although Adil’s study showed that hypertension and obesity were rare comorbidities in children with TIA, it is possible that of the 513 children analyzed, few had hypertension and obesity, but it is unclear whether those who did have them had a higher chance of getting TIA [1]. Our case did, however, support their study that those with prior TIA is a strong risk factor for recurrence.
After an initial ischemic stroke or TIA, secondary stroke prevention is important. It includes antiplatelet therapy, statins, and antihypertensives. The first line option in the absence of atrial fibrillation is aspirin, clopidogrel, or a combination of aspirin with dipyridamole [9]. Dual antiplatelet therapy is beneficial in the first three weeks but are typically switched to single antiplatelet [8]. Patients should also be started on high-dose statin, even if cholesterol levels are normal [8]. First line hypertensive drugs such as ACE inhibitors, calcium channel blockers, and diuretics are recommended for all patients with systolic blood pressure greater than 140/90 mmHg. Anticoagulants are indicated if the patient has atrial fibrillation; combinations of anticoagulants and antiplatelets drugs should be avoided [8]. Our patient is on Apixaban 5 mg, Aspirin 81 mg, Atorvastatin 40 mg, Lisinopril 10 mg, Rivaroxaban 20 mg, and Warfarin 5 mg. She’s on multiple anticoagulants, an antiplatelet, statin, and ACE inhibitor.
Warfarin is often prescribed to decrease the risk of stroke. It is a vitamin K antagonist that inhibits the vitamin K dependent coagulation factors II, VII, IX, and X. Vitamin K is essential for the activation of clotting factors by carboxylation, therefore inhibiting these factors will inhibit the formation of functionally active prothrombinase complexes and give an anticoagulant effect [2]. The therapeutic range of INR on warfarin is 2-3. Her INR ratio was 2.2, within range, meaning she was less likely to experience blood clotting while on the medication. In addition, aspirin at a daily dose of 75-162 mg or 500-1500 mg reduces longterm recurrence of stroke [5]. Adherence to diuretics like ACE inhibitors is beneficial to improve blood pressure long term and prevent secondary recurrence, and high-dose statin has shown to be protective. Although the patient was on multiple drug regimens, it is also unclear whether the patient was compliant with taking her medication [9]. A Norwegian study done in 2014 showed that in healthy men, a weight gain of >5 kg in early adult life was associated with increased risk of stroke and fit men who became unfit over 7 years had a greater stroke risk. Other factors such as her chronic history of hypertension and obesity could have a greater effect and role in TIA recurrence.
A study done in 2005 that looked at 2,416 patients who had an ischemic stroke showed that warning signs for ischemic stroke could be as evident as early as seven days before an attack [8]. 549 (23%) gave a history of a preceding TIA. 17% of TIA occurred on the day of stroke, 9% on the previous day, and 43% at some point during the 7 days prior to stroke. In our patient, she presented with signs three weeks prior to the attack [8]. It is unclear whether children present with warning signs longer before an attack or whether she had significant changes to her symptoms during the 7 days prior. Further investigation for timing could additionally help to better understand, diagnose, and treat children with TIA.
Conclusion
This case is unique due to the patient’s young age, presentation of lower extremity weakness for 3 weeks prior to transient ischemic attack (TIA) occurrence, and notable past medical history of cerebral artery occlusion with cerebral infarction, hyperlipidemia, and essential hypertension. Our report aims to highlight an alternate presentation of transient ischemic attacks, to ultimately help guide clinicians in prompt, efficient diagnosis of TIA, allowing for better patient management. Further investigation into TIA in younger populations and those with significant cardiovascular comorbidities would be beneficial.
Acknowledgement
None to state.
Conflict of Interest Statement
There are no conflicts of interest to disclose
References
1. Adil, M. M., Qureshi, A. I., Beslow, L. A., & Jordan, L. C. (2014). Transient ischemic attack requiring hospitalization of children in the United States: kids’ inpatient database 2003 to 2009. Stroke, 45(3), 887-888.
2. Kaithoju, S. (2015). Ischemic stroke: risk stratification, warfarin teatment and outcome measure. Journal of atrial fibrillation, 8(4), 1144.
3. Khare, S. (2016). Risk factors of transient ischemic attack: An overview. Journal of mid-life health, 7(1), 2-7.
4. Lehman. (2015). Predictors of Stroke After Transient Ischemic Attack in Children. American Heart Association Journal, 47, 88-93.
5. Lioutas, V. A., Ivan, C. S., Himali, J. J., Aparicio, H. J., & Leveille, T., et al. (2021). Incidence of transient ischemic attack and association with long-term risk of stroke. Jama, 325(4), 373-381.
6. Niu, P. P., Guo, Z. N., Jin, H., Xing, Y. Q., & Yang, Y. (2016). Antiplatelet regimens in the long-term secondary prevention of transient ischaemic attack and ischaemic stroke: an updated network meta-analysis. BMJ open, 6(3), e009013.
7. Panauganti. (2023). Transient Ischemic Attack. StatPearls.
8. Rothwell, P. M., & Warlow, C. P. (2005). Timing of TIAs preceding stroke: time window for prevention is very short. Neurology, 64(5), 817-820.
9. Tremonti, C., & Thieben, M. (2021). Drugs in secondary stroke prevention. Australian Prescriber, 44(3), 85-90.
10. Zhang, W. W., Cadilhac, D. A., Donnan, G. A., O'Callaghan, C., & Dewey, H. M. (2009). Hypertension and TIA. International Journal of Stroke, 4(3), 206-214.



