Case Report - (2023) Volume 8, Issue 2
Management of orbital diffuse large cell b lymphoma: a case report
Received Date: Jan 30, 2023 / Accepted Date: Feb 06, 2023 / Published Date: Feb 15, 2023
Abstract
Orbital lymphoma is a very rare location of large cell lymphomas. It accounts for 10% of orbital tumors. Proptosis is the most frequent sign, but it should be noted that the clinical presentation depends greatly on the initial site of the tumor. The diagnosis is suspected on clinical and radiological elements and confirmed by anatomopathologica exam.
Chemotherapy is the main treatment in the management of orbital lymphoma, often combined with radiation therapy and surgery.
We present a case of orbital diffuse large cell B lymphoma treated with chemothérapy with RCHOP protocol and external radiothérapy, and we discusse current recommendations from the literature
Keywords
Orbital Lymphoma, Radiothérapy, Tumor, Non-Hodgkin’s Lymphomas
Introduction
Non-Hodgkin’s lymphomas are monoclonal malignant proliferations of B or T lymphoid cells at different stages of maturation. Normally they usually develop in the lymph nodes,But we also find Primary extra-nodal localizations that represent less than one third of cases.
The orbit location is rare, it accounts for 10% of adult orbital tumors [1]. The median age is 60 years, and the clinical presentation include diplopia, decreased vision, proptosis, orbital mass, palpebral edema.
The diagnosis is based on clinical, radiological, histopathological and immunohistochemical examination [2].
Conventional treatment regimens are based on chemotherapy, possibly combined with radiotherapy +/- surgery, and close monitoring of the patient. Rituximab (Mabthera®) offers a significant alternative in the treatment of orbital non-hodgkin’s lymphoma [3]. Its efficacy has been documented especially in follicular NHL or in patients with relapses.
This case report shows treatment approach in a patient with right orbital diffuse large cell B lymphoma in our institution, and discusses current recommendations from the literature.
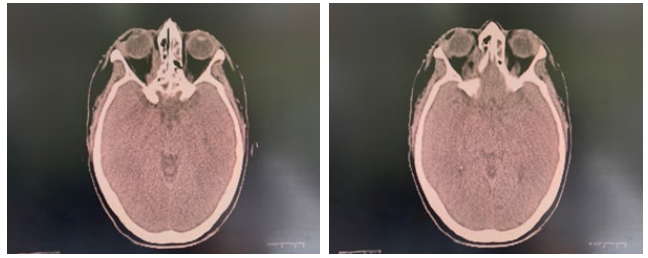
Figure 1: Post-chemothérapy orbital CT. focal thickening of the right medial oculomotor muscle.
Case Report
A 54-year-old Caucasian women with no personnal medical history, presented to the ophthalmology division of Rabat Hospital with a 1-year history of right-sided exophthalmos, painful and decreased visual acuity.
On examination, right-sided proptosis and restricted eye motility was noted. The patient’s visual acuity was 14/20, and visual fields were full. No facial or cervical lymphadenopathy was noted. Contrast-enhanced computed tomography (CT) scan of the head and orbits reveal locally advanced right retro-orbital tumor process (Figure 1).
The magnetic resonance imaging (MRI) scan of the head and orbits shows grade 1 exophthalmos with T2 hypersignal thickening of the right optic nerve, contrast-enhancing infiltration of the retroorbital fat and the optic nerve sheath. The PET-CT demonstrated an hypermetabolic rétro-orbital tumor process. A biopsy of this mass revealed a diffuse large cell B lymphoma.Neck, chest, abdomen and pelvis CT scans did not reveal any metastatic disease.
The patient was sent to oncology department for evaluation and was taken up for chemothérapy.Patient received R-CHOP (rituximab, doxorubicin, cyclophosphamide, vincristine and prednisolone) regimen, the total duration of chemotherapy was 3 months.
The response was evaluated through CT scan after the fourth, fifth, sixth cycle of the treatment, PET-CT and MRI after the last cycle of chémothérapy. The PET-CT showed a complete response after chemotherapy, MRI and CT demonstrated a focal thickening of the right medial oculomotor muscle with deviation of the optic nerve.
Then, consolidation Radiation was scheduled at weeks 13 to 14, and delivered dose was 36 Gy with 2 Gy per fraction. This dose was defined according to complete response after chemothérapy.
For planning radiation treatment,we use CT simulator (Siemens, Erlangen, Germany). The patient was immobilized in supine position with a thermoplastic mask 3 fixation points covering the head and neck. We acquired CT images from the head to the base of mandible with intravenous iodine contrast enhancement.The thickness of slice was 3 mm. after that, images were transferred to the Monaco treatment planning system version 5.11 (Elekta, Stockholm, Sweden) for delineation targets volumes. Then we coregistered CT images with pré-chemo PET-CT images in order to contour GTV pré-chemo.
The gross tumor volume (GTV) was defined before chémo, with the aim of better selecting the clinical target volume (CTV). For CTV, 5-mm margin was given around GTV to have high-risk CTV (CTV-HR), and was adjusted on the planning CT to restrictd natur al barriers such as bone and air, including the lacrimal fossa and the whole orbit. CTV was expanded by isotropic 5 mm margin to generate planning target volume (PTV). The dose prescription was 36 Gy at 2 Gy per fraction. OARs (cord, brainstem, optic chiasm, right eye, right lens, right retina, right optic nerve, temporal lobes and cochleae) were contoured. 3D conformationnel radiation was performed, using 3 fields with 6-MV photon, and 0.5-cm thick bolus (Figure 2).
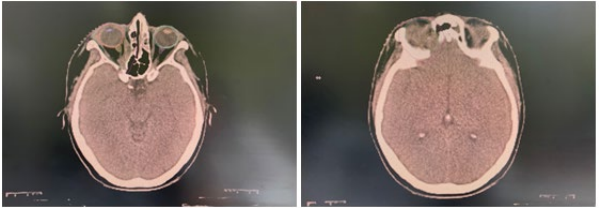
Figure 2: Axial views of the planning computed tomography scan showing the target volumes: pre-chemo GTV in yellow, CTV-HR and PTV-HR in green and pink, respectively
3-dimentionnal conformationnal plan was delivered using Elekta machine. Patient’s position was verified weekly by MV portal imaging prior to treatment. Patient was assessed weekly during radiation therapy in order to deliver treatment for any acute toxicities.
After the completion of treatment, patients were evaluated at 1 month for clinical exam and then every 3 months for first 2 years and then every 6 months. Clinical exam was realized at each follow-up,and CT/MRI every 6 months or earlier depending on symptoms. After 48 months follow-up, we didn’t note any local or distant recurrence, and the patient is still under regular follow-up until now.
Discussion
Orbital lymphoma is and accounted for 1% of all non-hodgkinien lymphoma [4]. It arises from the lacrimal gland, conjunctiva, ocular muscles and eyelid predominantly and accounts for 10% of adult orbital tumors.A majority (75% or more) of orbital lymphoma are low grade lymphoma and they have an indolent clinical course. If symptoms are present, They usually marked by exophthalmos which is the main symptom,ptosis,pain, and eye irritation, but exophthalmos is the most frequent sign in case of posterior development which is the case of our patient.
Data on Ann Arbor Staging was not available, so the classification depends on the type (lymphoma B or T), the grade and the stage of orbital lymphoma. Low-grade or low-malignancy lymphomas, also known as indolent, tend to develop very slowly. In this case, the prescription is rarely urgent. Indolent lymphomas usually respond well to treatment but they often relapse, sometimes repeatedly and after several years. Some indolent lymphomas can become aggressive.Then they require more intensive treatment. High-grade or intermediate and high malignancy NHLs are called aggressive. They tend to grow rapidly and require prompt treatment after diagnosis. However, they can be cure.
MRI and PET-CT if availables are a good exams for detection of small tumors and for evaluating tumor extension, but remain unable to differentiate between a malignant and benign tumor. The final diagnosis can only be made after pathological evaluation of a biopsy.
Among the lymphomas of the orbit and adnexa, 30 to 50% have a systemic disease [7], So an extension imaging, involving the superficial and deep lymph nodes, the bone marrow, the cerebrospinal fluid for high grade lymphoma is required,others imaging are necessary depending on clinical symptoms.
For treatment, Surgery for orbital lymphoma is usually not used, because of the difficulty of accessing the entire infiltrating mass for resection and to achieve complete surgery, with the concern of residual tumor [8].
The use of chemotherapy has not yet been clearly defined for localized disease [9]. In low grade Lymphoma, chemotherapy has no impact on survival improvement because of the indolent aspect of desease [10], and also because chemotherapy delays the radiation therapy which has a positif résult on survival. The chemotherapy regimens are COP, CHOP, or RCHOP with rituximab who shows a significant efficacy for nodal NH [11] and have been extrapolated for treating orbital lymphoma.In our case we have obtained a complete remission with this regimen (R-CHOP) and also a good tolerance to the treatment.
On the other hand, The good role of radiation was established when Esik et al. reported 42% local control with chemotherapy in comparison to 100% local control with radiotherapy [11]. In a retrospective study Avelis et al. Have shown 98% complete response after radiotherapy VS 100% complete response rate after combined chemo-radiation [12]. Radiation alone is recommended for low grade lymphoma, and combined modality with chemotherapy followed by radiothérapy are indicated for high grade, which may confer long term disease control and overall survival.
Due to the proximity of target volume to radiosensitive normal structures, advanced radiotherapy technologies are indicate for these patients, such as proton beam therapy and intensity modulated radiation therapy (IMRT), because of their high precision and the performance to deliver higher doses to the tumor and to spare adjacent organs at risk in order to reduce excessive late toxicity [13,14].
In our case, we have selected target volumes according to examinations, initial radiological extension of the disease and post-chemothérapy imaging. The GTV was delinated on the pre-chemo MRI and adjusted on the planning CT. Based on the international guidelines of the Danish Head and Neck Cancer group (DAHANCA), we contoured a CTV-HR as pre-chemo GTV plus a 5-mm margin, including the lacrimal fossa and the whole orbit along with the lateral orbital wall, superior orbital fissure, orbital roof. No elective nodes was délinated because there is no indication for prophylactic nodal irradiation in the litterature. PTV was created with 5-mm margin from the CTV to take into account daily setup variation.
However, the choice of radiation dose is still controversed, but the litterature shows a poor results in terms of local control and overall survival if the dose was lower than 30 Gy [15,16].
The dose depends on the degree of malignancy of the tumor, the grade and whether or not there is a systemic lymphoma. In case of isolated orbital lymphoma of low grade of malignancy, radiotherapy is indicated alone (30 Gy in 20 sessions in 4 weeks). For isolated orbital lymphoma of high grade of malignancy, we used induction chemotherapy followed by radiation dose of 30 Gy in case of complete response ,and 36 to 40 Gy for partial response. In our case(low grade lymphoma), We prescribed a dose of 30 Gy with standard fractionation to cover the micro- scopic disease after 6 cycles of chemothérapy(R-CHOP), which was consistent with data reported in the literature.
Cataract remains the major late toxicity, and Patients undergo radiation therapy may developp cataract [18] after doses of >16 Gy with a follow-up time of 3-9 years. Acute radiation effects are very minimal, especially with moderate dose treatment.
The largest cohort study O.M.AHMED et al. demonstrate that histology is an important predictor of survival, DLBCL is the worst prognosis (OS: 44.6% at 10 years) and MALT lymphoma conferring the best prognosis (OS: 64.9% at 10 years),
Conclusion
Orbital DLBCL is very rare with only few cases in our institution and in the litterature, it’s an uncommon sub-types of lymphomas. Prospective studies is not possible, they are restricted to retrospectives series of large databases or individuals cases reports. Optimal treatment is still debated. Conventional treatment regimens are based on chemotherapy, possibly combined with radiotherapy +/- surgery. Rituximab appears to be a significant aid in the treatment of indolent NHL, and Randomized studies are ongoing.
References
1. Shields JA, Shields CL, Scartozzi R (2004) Survey of 1264 patients with orbital tumors and simulating lesions : the 2002 Montgomery lecture, part 1. Ophthalmology 111:997-1008.
2. Madge SN, McCormick A, Patel I, Hatef E, Menon V, et al. (2010) Ocular adnexal diffuse large B-cell lymphoma: local disease correlates with better outcomes. Eye 24(6):954-961.
3. Hainsworth JD, Litchy S, Burris HA (2002) Rituximab as first-line and maintenance therapy for patients with indolent non-Hodgkin’s lymphoma. J Clin Oncol 20:4261-4267.
4. Fitzpatrick PJ, Macko S (1984) Lymphoreticular tumors of the orbit. Int J Radiat Oncol Biol Phys 10:333-3340.
5. Christopher E, Richard B Wilder, Chul S. Mark A, Fernando F, James D. Cox (2001) Clinical stage IEA – IIEA orbital lymphomas: outcome in the era of modern staging and treatment, Radiotherapy and Oncology 59:145-151.
6. Rykers K (2003) Orbital lymphoma: A simple treatment using electrons, Medical Dosimetry. 28:95-98.
7. Scott L, Timothy F. Kozelsky, James A. Garrity, Paul J. Kurtin, Jacqueline A. Leavitt, et al. (2001) Orbital lymphoma: radiotherapy outcome and complications, Radiotherapy and Oncology 59:139-144.
8. EÌsikO, IkedaH, MukaiK,KanekoA (1996) A retrospective analysis of different modalities for treatment of primary orbital non–Hodgkin’s lymphomas. Radiother Oncol 38(1):13-18.
9. Letschcrt JGJ, Gonz&s Gonzales D, Oskam J, Koomneef L, van Dijk JDP, et al. (1991) Results of radiotherapy in patients with stage I orbital non-Hodgkin’s lymphoma. Radiother. Oncol 22:36-44.
10. Letschert JG, Gonazales Gonazales D, Oskam J, Koornneef L, van Dijk JD, et al. (1991) Results of radiotherapy in patients with stage-I orbital non-Hodgkin’s lymphoma. Radiother Oncol 22:36-44.
11. Coiffier B, Lepage E, Briere J, Herbrecht R, Tilly H, et al. (2002) CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large B-cell lymphoma. N Engl J Med 346:235-242.
12. Avile Ìs A, Neri N, Calva A, Huerta-Guzma Ìn J, Cleto S, et al. (2006) Addition of a short course of chemotherapy did not improve outcome in patients with localized marginal B-cell lymphoma of the orbit. Oncology 70:173-176.
13. Purdy JA, Low D, Chao KS (2004) Principles and practice of radiation oncology. Three-dimensional conformal radiation therapy: physics, treatment planning and clinical aspects. Philadelphia: Lippincott Williams and Wilkins 283-313.
14. Mohan R, Low D, Chao KS (2004) Principles and practice of radiation oncology. Intensity-modulated radiation treatment planning, quality assurance, delivery and clinical application. Philadelphia: Lippincott Williams and Wilkins 31-36.
15. Kamath SS, Marcus Jr RB, Lynch JW, Mendenhall NP. The impact of radiotherapy dose and other treatment-related and clinical factors on in-field control in stage I and II non- Hodgkin’s lymphoma. Int J Radiat Oncol Biol Phys 1999;44:563-568.
16. Foster SC, Wilson CS, Tretter PK (1971) Radiotherapy of the primary lymphoma of the orbit. Am J Roentgenol Radium Ther Nucl Med 111:343-349.
17. Knowles DM, Jakobiec FA (1992) Malignant lymphomas and lymphoid hyperplasias that occur in the ocular adnexa (orbit, conjunctiva, and eyelids). In: Neoplastic Hematopathology, pp. 1009-1046. Editor: D.M. Knowles. Williams and Wilkins, Baltimore.
18. Henk JM, Whitelocke RAF, Warrington AP, Bessel EM (1993) Radiation doseto the lens and cataract formation. Int J Radiat Oncol Biol Phys 25:815-820
Copyright: © 2025 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.



