Review Article - (2024) Volume 9, Issue 12
Choice of Fluid in the Acute Management of Diabetic Ketoacidosis
Received Date: Nov 25, 2024 / Accepted Date: Dec 03, 2024 / Published Date: Dec 28, 2024
Abstract
Diabetic ketoacidosis (DKA) is a potentially life-threatening complication which occurs mainly in patients with type 1 diabetes, but it is also not uncommon in type 2 diabetics. In the United States, it accounts for 30 admissions per 1000 diabetic patients every year, with a mortality of 1-5% [1]. DKA is characterized by high anion gap metabolic ketoacidosis, hyperglycemia and ketonuria. Stress events including infection, non-compliance to medications are triggers of DKA. Fluid administration to address hypovolemia and ensure adequate kidney perfusion, along with insulin and electrolytes replacement are cornerstones of management. Currently, in terms of fluid resuscitation, the Joint British Diabetes Society (JBDS) guideline and the American Diabetes Association (ADA) recommends the use of normal saline (NS) [2, 3]. The choice of fluid is debatable as no definite superiority of one type of fluid over another is clearly demonstrated. However, studies have shown improvement in certain clinical outcomes, for instance, quicker resolution of DKA when balanced crystalloid fluids are used [4]. This essay mainly reviews the currently available evidence on the differences in clinical outcomes with respect to choice of crystalloids in treating DKA patients.
Keywords
Diabetic ketoacidosis, American Diabetes Association, Ketoacidosis, Hyperglycemia, Ketonuria
Introduction
Diabetic ketoacidosis (DKA) is a potentially life-threatening complication which occurs mainly in patients with type 1 diabetes, but it is also not uncommon in type 2 diabetics. In the United States, it accounts for 30 admissions per 1000 diabetic patients every year, with a mortality of 1-5% [1].
DKA is characterised by high anion gap metabolic ketoacidosis, hyperglycemia and ketonuria. Stress events including infection, non-compliance to medications are triggers of DKA. Fluid administration to address hypovolemia and ensure adequate kidney perfusion, along with insulin and electrolytes replacement are cornerstones of management. Currently, in terms of fluid resuscitation, the Joint British Diabetes Society (JBDS) guideline and the American Diabetes Association (ADA) recommends the use of normal saline (NS) [2, 3]. The choice of fluid is debatable as no definite superiority of one type of fluid over another is clearly demonstrated. However, studies have shown improvement in certain clinical outcomes, for instance, quicker resolution of DKA when balanced crystalloid fluids are used [4], This essay mainly reviews the currently available evidence on the differences in clinical outcomes with respect to choice of crystalloids in treating DKA patients.
Discussion
Regarding the composition of different fluids (Table 1), the commonly used NS is characterised by equal concentrations of sodium and chloride. The supraphysiological concentration of chloride (154mEq/L) in NS, when compared to human plasma and lactated ringer’s (LR), contributes to a higher risk of iatrogenic non-anion-gap hyperchloremic metabolic acidosis, particularly when large volumes are used. This is due to the lowering of other primary plasma anions, for example, bicarbonate, to maintain electroneutrality, leading to acidosis. The excess chloride further worsens the existing acidosis and masks the resolution of acidosis [5]. In contrast, the chloride concentration in balanced crystalloids is similar to human plasma and the presence of anions (sodium lactate in LR and acetate in Plasma-Lyte A) has an neutralising/alkalising effects due to its metabolism into bicarbonate. All these factors contribute to the quicker resolution of ketoacidosis.
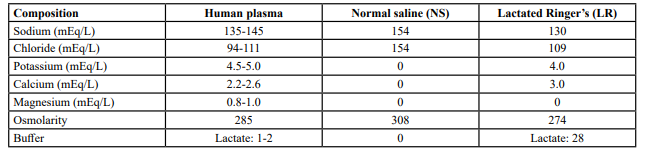
Table 1: Composition of human plasma, Normal Saline and Lactated Ringer’s [6]
A number of studies have compared the use of NS to balanced crystalloids in the acute management of DKA patients in the adult population. Firstly, a double blinded randomised controlled trial published in QJM studied whether LR is superior to NS in resolution of acidosis in DKA patients by measuring the time to pH normalisation (pH of 7.32) as the primary endpoint [7]. Other clinical endpoints include, time to achieve serum glucose of 14 mmol/l and time to DKA resolution. DKA resolution was initially defined as venous pH > 7.3, serum bicarbonate ≥18 mmol/l and blood glucose <11.1 mmol/l by the American Diabetes Association (ADA) in 2006. Then, the ADA criteria was changed in 2009–Serum bicarbonate level was changed from ≥18 mmol/l to ≥15 mmol/L and anion gap ≤12 mEq/L was added as one of the three considerations, apart from pH and bicarbonate level. Therefore, in this study, serum bicarbonate levels and electrolytes were not taken into account initially as per 2009 ADA resolution criteria. As a result, the anion gap could not be fully assessed. The median time to reach a pH value ≥7.32 was 683 mins for the NS group and 540 mins for the LR group, yet the difference is not statistically significant (P=0.251). The difference in time taken for DKA resolution (based on ADA criteria) was also statistically insignificant (P=0.758). Meanwhile, administration of LR demonstrated a delay in the fall in blood glucose level (P=0.044), which could be explained by the underlying physiological mechanism of lactate metabolism [8]. The study design limits the comparability of fixed time measurements as the timing of blood sampling was progressively shorter in order to achieve the time of DKA resolution as accurately as possible. Small sample size resulting in low power of the study was another limitation because only data from 54 patients were analysed.
Secondly, another subgroup secondary analysis was conducted as part of two randomised clinical trials—Saline [9]. Against Lactated Ringer’s or Plasma-Lyte in the Emergency Department (SALT-ED) and the Isotonic Solutions and Major Adverse Renal Events Trial (SMART). Of the 172 DKA patients involved, 78 of them were randomly assigned to the NS group and 94 to the balanced crystalloid group. In the balanced crystalloid group, LR was mainly administered (96.9%) whilst the remaining 3.1% was given Plasma-Lyte A. The primary endpoint was time to DKA resolution, as defined by the American Diabetes Association [3]–plasma glucose7.3 and anion gap ≤12mEq/L. According to the unadjusted cumulative incidence and multivariable proportional hazards model, there is an absolute reduction of around 4 hours and a relative reduction of approximately 20-30% in time for DKA resolution based on median values. Patients receiving balanced crystalloids took a shorter median time to achieve DKA resolution (13.0 hours) when compared with the saline group (16.9 hours), with a P value of 0.04.
Median time to discontinuation of insulin infusion was also shorter in the group receiving balanced crystalloids (9.8 hours) than the group receiving NS (13.4hours), with a P value of 0.03. Additionally, higher levels of bicarbonate and lower levels of chloride were seen in the group receiving balanced crystalloids. Meanwhile, the use of saline was not associated with superior outcomes e.g. major adverse kidney events, the need for mechanical ventilation, when compared to balanced crystalloids. Since this is a subgroup analysis of the SALTED and SMART trial, the number of patients enrolled were determined by the sample size of the primary study, thus the power was not calculated specifically for this study. Data on race was based on self-report and only classified as white or nonwhite. Therefore, samples are not extensive enough to address the variability in other populations and rare clinical outcomes. For patients not fully meeting the ADA laboratory criteria for DKA resolution before discharge, time of hospital discharge was considered as resolution time. This limits the generalizability of the findings. Moreover, comparison between LR and Plasma Lyte was inadequate as it was the physician’s choice to select between these two balanced crystalloids, which then bring out another factor of bias–the unblinded intervention, along with variables that are not addressed, including insulin dosing and volume of fluid given.
Thirdly, RINSE-DKA is a recently published cohort study which included 771 patients (mostly white) and compared the use of LR and NS in the management of DKA [10].
Primary outcome was time to high anion gap metabolic acidosis resolution. Results showed that LR was associated with a shorter duration (14.3 hours) for HAGMA resolution compared to NS (16.7 hours) (P17mEq/L and an anion gap<18 mEq/L) is different from the ADA criteria. Of those who satisfied the ADA resolution criteria (86.8% in NS group and 91% in LR group), there was no statistically significant difference between the NS group and LR group, in terms of time to DKA resolution (P=0.184). This study also included a subgroup analysis of patients with DKA and sepsis/septic shock, which revealed a lower incidence of nonanion-gap metabolic acidosis in the LR group compared with the NS group (67.5% VS 36.7%, P=0.004), in addition to faster resolution of HAGMA (P<0.001).
Nevertheless, there were no significant differences demonstrated in other clinical outcomes between the two groups, for example, hyperchloremia or nonanion gap metabolic acidosis. In terms of limitation, volume of fluid given and the significant use of more sodium bicarbonate in the NS group (P=0.033) are variables that are not interpreted.
Implications
The sample sizes of the readily available studies are not large enough to allow a more precise estimate of the treatment effect and provide a more accurate result. The use of various criteria, for instance, not strictly adhering to ADA criteria, limits the validity and comparability of different findings. Multiple studies have adopted the time to DKA resolution as a clinical outcome. This is because faster resolution of DKA prevents electrolyte abnormalities and its associated complications. As most of the available studies present data on the Caucasian population (or not mentioned), studies in other ethnicities are warranted. There is an ongoing systematic review which involves both the adult and pediatric population with either DKA or HHS (hyperosmolar hyperglycemic state) [11]. Endpoints include clinical outcomes(overall mortality, development of altered conscious level, length of hospital stay, etc), biochemistries, and endocrine-specific outcomes (pH, anion gap, electrolytes, etc) in patients who are administered 0.9% saline as compared to other buffered and non-buffered crystalloid fluids, instead of mainly LR. The wider inclusion and assessment criteria could potentially add value to the currently available literature.
Conclusion
Overall, with the emerging evidence, the use of balanced crystalloids over normal saline is safe and may be beneficial in optimising the management of DKA patients.
References
1. Benoit, S. R., Zhang, Y., Geiss, L. S., Gregg, E. W., & Albright, A. (2018). Trends in diabetic ketoacidosis hospitalizations and in-hospital mortality — United States, 2000–2014. MMWR. Morbidity and Mortality Weekly Report, 67(12), 362–365.
2. Dhatariya, K. K. (2022). The management of diabetic ketoacidosis in adults—an updated guideline from the joint British diabetes Society for Inpatient Care. Diabetic Medicine, 39(6).
3. Kitabchi, A. E., Umpierrez, G. E., Miles, J. M., & Fisher, J. N. (2009). Hyperglycemic crises in adult patients with diabetes. Diabetes care, 32(7), 1335–1343.
4. Self, W. H., Evans, C. S., Jenkins, C. A., Brown, R. M., Casey, J. D., Collins, S. P., Coston, T. D., Felbinger, M., Flemmons, L. N., Hellervik, S. M., Lindsell, C. J., Liu, D., McCoin, N. S., Niswender, K. D., Slovis, C. M., Stollings, J. L., Wang, L., Rice, T. W., & Semler, M. W. (2020). Clinical effects of balanced crystalloids vs saline in adults with diabetic ketoacidosis. JAMA Network Open, 3(11).
5. Taylor, D., Durward, A., Tibby, S. M., Thorburn, K., Holton, F., Johnstone, I. C., & Murdoch, I. A. (2006). The influence of hyperchloraemia on acid base interpretation in diabetic ketoacidosis. Intensive Care Medicine, 32(2), 295–301.
6. Dunn, B. K., Coore, H., Bongu, N., Brewer, K. L., Kumar, D., Malur, A., & Alkhalisy, H. (2024). Treatment challenges and controversies in the management of critically ill diabetic ketoacidosis (DKA) patients in Intensive Care Units. Cureus, 16(9): e68785
7. Van Zyl, D. G., Rheeder, P., & Delport, E. (2011). Fluid management in diabetic-acidosis--Ringer’s lactate versus normal saline: A randomized controlled trial. QJM, 105(4), 337–343.
8. Hartmann, A. F., & Senn, M. J. (1932). Studies in the metabolism of sodium r-lactate. II. Response of human subjects with acidosis to the intravenous injection of sodium r-lactate. The Journal of Clinical Investigation, 11(2), 337- 344.
9. Wesley H. Self, M. (2020a). Clinical effects of balanced crystalloids vs saline in adults with diabetic ketoacidosis. JAMA Network Open, 3(11):e2024596
10. Jamison, A., Mohamed, A., Chedester, C., Klindworth, K., Hamarshi, M., & Sembroski, E. (2024). Lactated Ringer’s versus normal saline in the management of acute diabetic ketoacidosis (rinseâ?dka). Pharmacotherapy: The Journal of Human Pharmacology and Drug Therapy, 44(8), 623–630.
11. Gershkovich, B., English, S. W., Doyle, M.-A., Menon, K., & McIntyre, L. (2019). Choice of crystalloid fluid in the treatment of hyperglycemic emergencies: A systematic review protocol. Systematic Reviews, 8(1).
Copyright: © 2025 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.



